Academic Frontiers | Research on the Use of High-Frequency Spinal Cord Stimulation to Relieve Muscle
From: News Date: 2026-01-26 16:24Spasticity is secondary to a variety of diseases, including brain injury, spinal cord injury, etc., and the symptom is increased muscle tone. Although there are many medical treatments for this symptom, the clinical effects of conventional drug and surgical treatment are often insufficient, and conventional non-invasive electrical stimulation cannot target deep muscle groups. In recent years, epidural spinal cord stimulation (SCS) has shown good efficacy in patients with spinal cord spasticity (such as spinal cord injury) and brain-derived spasticity (such as cerebral palsy, stroke, multiple sclerosis, etc.)[1]. With the progress of research on SCS to relieve spasticity, high frequency spinal cord stimulation (HF-SCS) has been explored in recent years to improve spasticity and muscle tone. This article mainly summarizes the relevant research.
I.1200Hz high-frequency SCS stimulation relieves spasticity in patients with spinal cord injury and promotes recovery of walking ability.
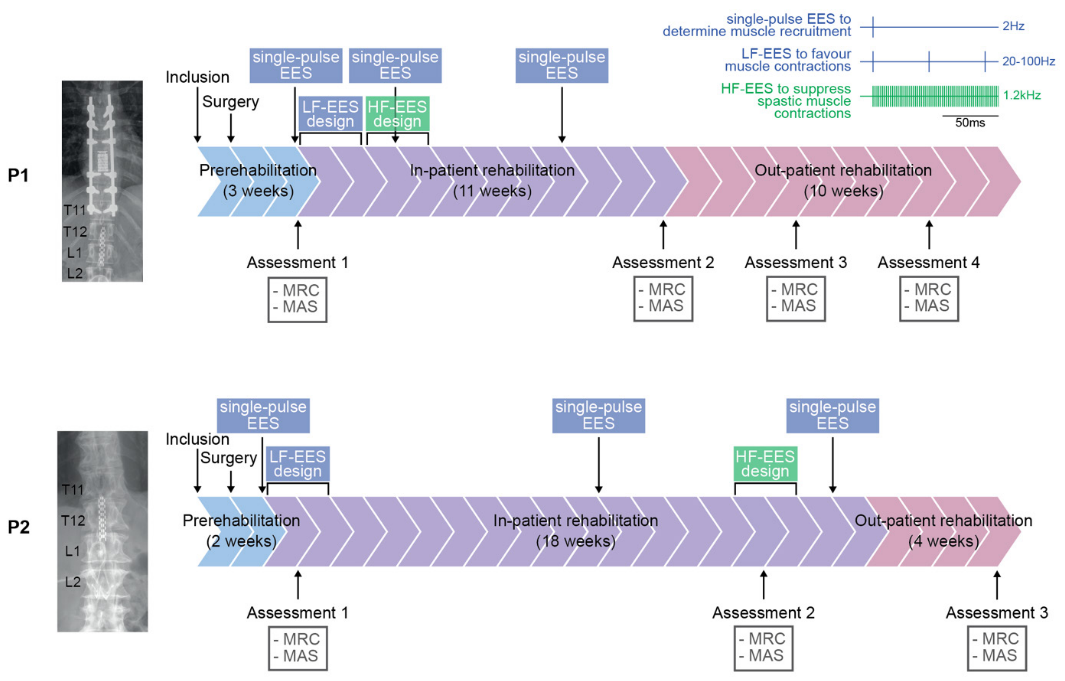
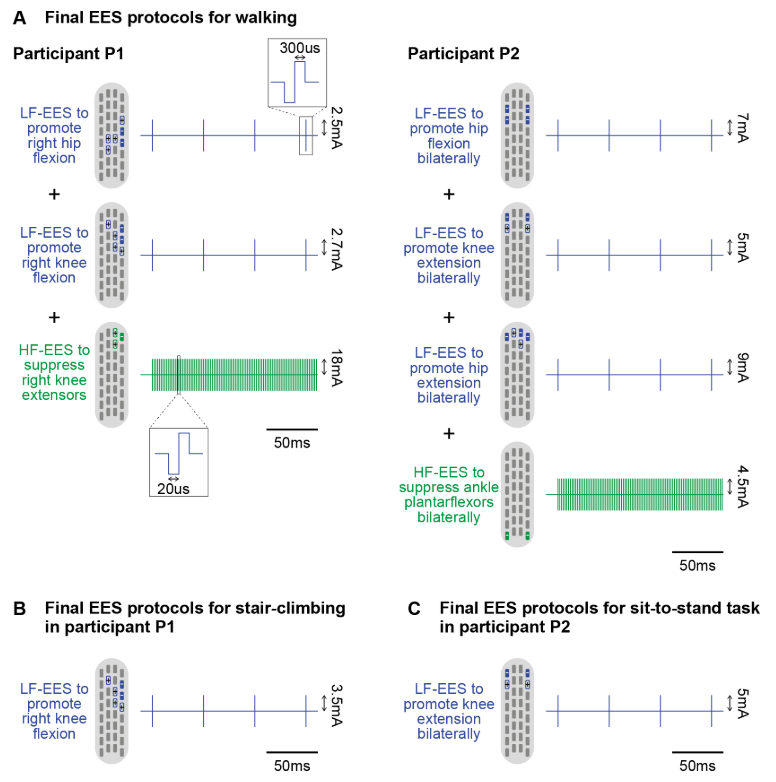
About 70% of patients with thoracic spinal cord SCI have spasticity such as high muscle tone and clonus. The side effects of drugs and destructive surgical methods are obvious. In order to verify the effect of 1200Hz HF-SCS in inhibiting spasticity and promoting lower limb movement, in 2025, Simone et al. implanted the SCS system in two patients with incomplete thoracic spinal cord injury with obvious spasticity and muscle synergistic contraction, covering the T11-T12 vertebral bodies. Based on the theoretical basis of "kilohertz block" in the peripheral nerve field, personalized high frequency epidural electrical stimulation (HF-SCS) parameters were designed. The first comprehensive program of HF-SCS combined with traditional low frequency epidural electrical stimulation (LF-SCS) and enhanced rehabilitation training was evaluated to improve the motor function of patients [2]. The low frequency stimulation parameters were 20-100Hz (mainly 40Hz) and 300μs, and the amplitude was set between the sensory threshold (causing the patient to perceive) and the motor threshold (causing involuntary muscle contraction). The high-frequency stimulation parameters are 1.2kHz and 20μs, with the amplitude set to the maximum value that the patient can tolerate.
methods
1.1 LF-SCS recruits major muscle groups of the lower limbs
To determine muscle recruitment under different electrode contact configurations, single-pulse epidural electrical stimulation (SCS) with a pulse width of 300 μs, a frequency of 2 Hz, and increasing amplitude was applied to the patient while supine. Muscle recruitment was assessed using the normalized peak-to-peak amplitude values of each pulse in surface electromyography (EMG). Electrode contact configurations that selectively activated the major muscle groups of the right leg (the side with the most severe motor impairment) in subject P1 and bilateral lower limbs in subject P2 were selected. High functional selectivity was achieved for the hip flexors [rectus femoris (RF)], hip extensors [gluteus maximus (GLmax)], knee flexors [biceps femoris (BF) and semimembranosus (SM)], knee extensors [vastus lateralis (VL)], and ankle plantar flexors [gastrocnemius (GM)].
For subject P1, an electrode contact configuration targeting right leg movement was selected. LF-SCS applied using these electrode contacts enhanced EMG activity and joint angles during various movements, including knee flexion (biceps femoris BF and semimembranosus SM), triple leg flexion (rectus femoris RF, biceps femoris BF, semimembranosus SM, and tibialis anterior TA), as well as knee extension (vastus lateralis VL), ankle plantar flexion (gastrocnemius medial head GM), hip extension (gluteus maximus GLmax), ankle dorsiflexion (tibialis anterior TA), and hip flexion (rectus femoris RF) (Video 1).
LF-EES restores or enhances autonomous movement of single and multiple joints
1.2 Designing the HF-SCS protocol to inhibit spastic muscle contractions
For subject P1, a HF-SCS program targeting the knee extensors (vastus lateralis, VL) improved the synergistic contraction of the right knee extensors and flexors during walking. A similar HF-SCS program was designed for subject P2 to inhibit excessive activity of the right knee extensors, thereby training voluntary knee flexion in the prone position. Subject P2 exhibited moderate pathological synergistic contraction of the ankle plantar flexors (gastrocnemius), which we hypothesized might limit active dorsiflexion of the foot, thus affecting gait performance. Therefore, an HF-SCS program was designed to inhibit the plantar flexor muscles of both legs.
1.3 HF-SCS inhibits stretch reflex and ankle clonus
In subject P1, who had more severe hyperreflexia, the effect of HF-SCS was not immediately apparent after stimulation began, but rather took several minutes to take effect. In subject P2, who had milder hyperreflexia, the application of HF-SCS immediately caused a significant reduction in reflex amplitude and the disappearance of leg rebound (Video 2). Furthermore, similar to patient P1, compared to the unstimulated state, HF-SCS resulted in a more significant decrease in reflex amplitude in subsequent tests, indicating that its time-dependent enhancement effect also existed in P2.
In subject P2, the inhibitory effect of HF-SCS on bilateral ankle clonus was also investigated. The effects of applying HF-SCS and low-frequency epidural electrical stimulation (LF-SCS) with the same electrode contact configuration (targeting the medial head of the gastrocnemius muscle) were compared using a rapid dorsiflexion stretch test (Video 2). The experiment showed that while both HF-SCS and LF-SCS could inhibit ankle clonus, LF-SCS caused a rebound in muscle contraction (manifested as a peak ankle kinematic response), leading to a reduction in the range of motion of both legs.
The efficacy of HF-EES on subject P2
1.4 HF-SCS reduces fatigue and improves functional task performance
For subject P1, the combined HF-SCS and LF-SCS protocol more effectively improved hip flexion kinematics during treadmill walking. In the stair-climbing task, the swing-phase hip kinematic parameters produced by HF-SCS were not statistically different from those produced by closed stimulation or LF-SCS stimulation, but HF-SCS produced higher repeatability results. HF-SCS was also associated with reduced variability in electromyographic activity (EMG) of task-independent muscles, including knee extensors and flexors during the swing phase of gait. Reduced EMG activity in inactive muscles may indicate reduced energy waste, thus reducing fatigue. Finally, in the sit-to-stand task, applying HF-SCS to the primary muscle group (knee extensors) did not reduce EMG activity or affect task success rate.
In subject P2, the facilitating effects of high-frequency epidural electrical stimulation (HF-SCS) on single-joint movement and walking function were evaluated. After applying HF-SCS to the ankle plantar flexors, patients showed a significant improvement in voluntary bilateral ankle dorsiflexion (Video 2). Furthermore, the HF-SCS targeting the knee extensors enhanced voluntary knee flexion in the prone position. In the same specific position, the LF-SCS, with electrode contacts targeting the knee flexors, actually reduced the range of motion compared to no stimulation. Finally, by combining the HF-SCS protocol targeting the right gastrocnemius (GM) with the LF-SCS protocol specifically designed for walking, a significantly shorter completion time for the 10-meter walk test was observed compared to no stimulation or LF-SCS alone. In the 6-minute walk test (repeated twice on different days), as the task progressed, combined stimulation with LF-SCS and HF-SCS increased ankle dorsiflexion angle more than LF-SCS alone.
1.5 A rehabilitation program integrating LF-SCS and HF-SCS can improve motor function
Subject P1's rehabilitation treatment improved his lower limb voluntary motor function (Video 3). Assessments using the MRC muscle strength scale and dynamometer showed improvement in multiple muscle groups. The subject also showed improvement in the 6-minute walk test and timed stand-up walking test. Modified Ashworth Scale (MAS) data showed improvement in spasticity of the hip extensors (GLmax) and primarily the ankle plantar flexors (GM).
Subject P2's rehabilitation process also showed a longitudinal improvement trend (Video 4). Over time, both his lower limb MRC scores and muscle strength improved. The 6-minute walk test showed a steady increase in walking distance. The patient's performance in the 10-meter walk test and sit-to-stand test also continued to improve. Compared to the off state, electrical stimulation (SCS) produced superior efficacy indicators at each assessment time point.
Long-term neurorehabilitation effects of subject P1
Long-term neurorehabilitation effects of subject P2
1.6 Long-term neurorehabilitation effects of subject P2
2. 10kHz ultra-high frequency SCS effectively relieves pain and significantly alleviates muscle spasms.
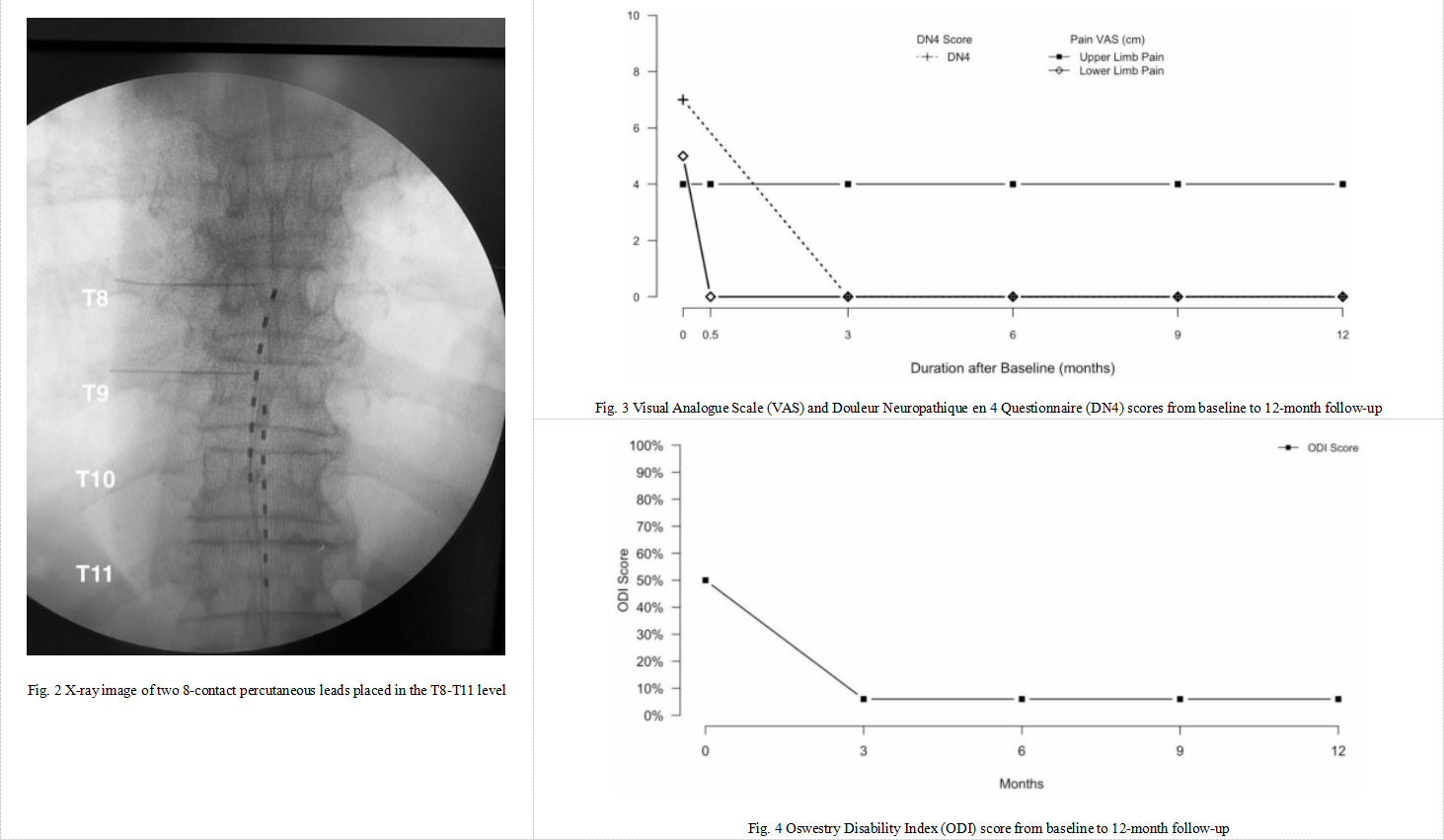
10kHz ultra-high frequency SCS (HF10 SCS) has good analgesic effect and is not affected by the patient's position, and is widely used in the treatment of chronic pain. In 2024, Gazzeri et al. published a single-case study of HF10 SCS in the treatment of spastic quadriplegia secondary to cervical spinal cord softening [3]. The patient was a 66-year-old male diagnosed with C3-C6 spinal cord softening with spastic quadriplegia, who had previously undergone decompression laminectomy. Symptoms included burning pain, spasm, and paresthesia in the upper and lower limbs, and severely limited walking ability. Due to the formation of epidural scar tissue after the patient's C3-C6 laminectomy, electrodes could not be implanted at the cervical level, and ultimately only two electrodes were implanted in the T8-T11 segment. The stimulation parameters were 10kHz frequency, 30μs pulse width, and amplitude of 0.5-3.5mA, and the contact point position and intensity were optimized based on patient feedback. After treatment, the patient's lower limb pain was completely relieved (VAS=0), the DN4 score decreased from 7 to 0, functional impairment was significantly reduced, ODI improved from 50% to 6%, lower limb spasticity completely disappeared, muscle tone was significantly reduced, and the patient regained the ability to walk independently, but upper limb symptoms did not improve. This study is the first case of using 10 kHz SCS to treat spastic quadriplegia, resulting in spasticity recovery and motor improvement. As a stimulation without tingling sensation, this therapy overcomes the limitations of traditional low-frequency SCS in spinal cord and cerebrospinal fluid shunting and target tissue recruitment. It not only produces pain relief but also significantly improves motor function and spastic symptoms, providing a new direction for the comprehensive treatment of such patients. 10 kHz achieves analgesia through desensitization and inhibitory reconstruction of the spinal dorsal horn and related networks, while simultaneously achieving kilohertz blockade to prevent muscle spasticity.
3. Summary
The implantation site of SCS electrodes for treating muscle tone problems depends mainly on the treatment target. For quadriplegia and upper limb spasticity, the electrode implantation site is C2-T1, and the lower cervical spine is preferred in clinical practice. The abundant interneurons in the cervical enlargement can be used to reconstruct synaptic connections and promote the recovery of upper limb motor function. For lower limb spasticity and gait disorders, the electrode implantation site is T10-L2, and the preferred clinical target is the thoracolumbar junction, which can induce motor patterns by stimulating the lumbosacral enlargement [1].
Traditional stimulation protocols mostly use the LF-SCS mode, with parameters generally selected as 2-5 mA current and frequency below 350 Hz [1]. Existing studies on HF-SCS have demonstrated to some extent the effectiveness of this mode in improving spasticity and muscle tone, with parameters selected as a high-frequency mode of 1.2 kHz and an ultra-high-frequency stimulation mode of 10 kHz. However, due to the limited sample size, the conclusions still need extensive verification.
References
[1] JUNG Y, BREITBART S, MALVEA A, et al. Epidural Spinal Cord Stimulation for Spasticity: a Systematic Review of the Literature[J/OL]. World Neurosurgery, 2024, 183: 227-238.e5.
[2] ROMENI S, LOSANNO E, EMEDOLI D, et al. High-frequency epidural electrical stimulation reduces spasticity and facilitates walking recovery in patients with spinal cord injury[J/OL]. Science Translational Medicine, 2025, 17(780): eadp9607.
[3] GAZZERI R, GALARZA M, OCCHIGROSSI F. Motor improvement and spasms recovery with high-frequency 10 kHz spinal cord stimulation in a patient with spastic tetraparesis: beyond pain relief[J/OL]. European Spine Journal, 2024, 33(12): 4750-4756.
Contact